Abstract
Background:
Myeloproliferative neoplasms (MPNs) are a heterogenous group of chronic hematologic malignancies that lead to morbidity and early mortality primarily due to thrombotic complications and progression to acute leukemia. Clinical and mutational risk factors have been demonstrated to predict outcomes in patients with MPNs, and are used commonly to guide therapeutic decisions (including allogenic stem cell transplant) in myelofibrosis. Adolescents and young adults (AYA, age ≤45 years) comprise less than 10% of all MPN patients, and have unique clinical and therapeutic considerations. The prevalence and clinical impact of somatic mutations implicated in myeloid disease has not been extensively reported in this population.
Methods:
We conducted a retrospective review of patients evaluated at the Princess Margaret Cancer Centre (PMCC) between 1/1/2000 and 30/6/2021 for an MPN diagnosed at ≤45 years of age. Diagnoses were defined per 2016 WHO criteria using available information on chart review. Targeted mutational profiling of clinically relevant myeloid genes (49- or 54-gene panel) was previously performed on peripheral blood or bone marrow samples. Pathologic variants in ASXL1, EZH2, IDH1/2, SRSF2, and U2AF1 Q157 were categorized as high-molecular risk (HMR). Categorical variables were compared using the χ2 test. Kaplan-Meier method was used for overall survival (OS) and time-to-event analysis; and were compared using the log-rank test.
Results:
A total of 237 patients with initial MPN diagnosis ≤45 years were included in the study, with median age of diagnosis 35 (range 12-45) years. MPN diagnosis included: essential thrombocythemia (ET, n=100), polycythemia vera (PV, n=75), prefibrotic primary myelofibrosis (Pre-PMF, N=29), overt PMF (n=24), and MPN-unclassifiable (MPN-u, n=9). Driver mutation data, available for 230 (97%) patients, were: JAK2 in 134 (56%), CALR in 70 (30%), MPL in 6 (2.5%), and triple-negative in 20 (8.4%) patients.
Median follow-up was 10.2 (range 0.3-45.3) years, and median OS was the shortest for those with overt PMF (21.4 years, P=0.001) compared with PV (33.0 years), ET (31.4 years), and Pre-PMF (median OS not reached). No difference in OS based on driver mutation was observed. Progression to a secondary MF (SMF) was observed in 84 patients (47% of ET, 48% of PV) after median time of 19.7 years; with no difference in time to progression due to diagnosis or driver mutation. Progression to an accelerated/blast phase (AP/BP) was observed in 26 patients and was associated with diagnosis of overt PMF (P=0.04) and triple-negative for driver mutation (P=0.04).
Thrombosis was observed in 61 (26%) patients: 34 (14%) patients with thrombosis prior to/concurrent with MPN diagnosis, and 34 (14%) patients with thrombosis after diagnosis. Splanchnic circulation thrombosis was found in 29 (12%) patients and 12 (5%) patients had Budd-Chiari syndrome. Portal hypertension was reported in 37 (16%) patients. CALR type-2 mutations were associated with the lowest frequency of total thrombosis (4%, P=0.02); while JAK2 had the greatest frequency of splanchnic circulation clot (17%, P=0.03).
Targeted mutational profile was obtained for 202 (85%) of patients: 135 samples obtained during the initial disease phase, 51 samples during a secondary MF phase, and 16 during an AP/BP. For the patients with mutation analysis in the initial phase, 24 pathogenic mutations were observed in 16 (12%) patients including 3 patients with HMR mutations. The most frequent mutations observed were in TET2 (n=12, 9%), and ASXL1 (n=3, 2%). Presence of additional mutations did not predict OS, AP/BP progression, or thrombosis; though TET2 mutation was associated with shorter time to SMF progression in patients with PV/ET (P=0.002). Additional mutations were found in 34/51 (67%) patients in whom mutational analysis was first performed during a SMF disease phase.
Conclusions:
A long-term follow-up study of a large molecularly annotated cohort of AYA patients with MPN demonstrated excellent long-term survival for these patients. Overt PMF is associated with the lower OS and higher risk of AP/BP transformation. Thrombotic complications including splanchnic circulation thrombosis are frequent complications. Mutations aside from JAK2/MPL/CALR are uncommon and HMR mutations rare in the initial phase of MPN in the AYA population.
Gupta: Roche: Consultancy; Pfizer: Consultancy; AbbVie: Consultancy, Honoraria; BMS-Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Sierra Oncology: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Incyte: Honoraria, Research Funding; Constellation Pharma: Consultancy, Honoraria. Maze: Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene-BMS: Honoraria; Takeda: Research Funding; PharmaEssentia: Research Funding; Kronos Bio: Research Funding.
Author notes
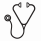 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract